Carbon Capture & Storage
Increasing CO2 site safety and storage capacity
ESal's RightWater® Technology has the capability to solve for the following:
- Increase safety of CO2 storage
- Perform full-field reservoir simlations
- Integrated modelling (Geochemical and Geomechanical)
- Flow assurance
- CO2 Storage Development
- Increase the target reservoir CO2 capacity
- Develop a monitoring, measurement, and validation plan
- Review and screen sites and maturation studies
- Review existing models
- Structural Interpretation
Lock down more CO2 in less space with ESal
Carbon capture and storage (CCS) is the capture of CO2 emissions from industrial sources and injection into deep, porous underground formations, rather than release into the atmosphere. Capture and injection are developed technologies that are being constantly improved with the goal of trapping CO2in the ground for generations. There are 4 mechanisms that trap CO2: stratigraphic (cap rock), residual or capillary trapping, solubility and mineralization. Capillary trapping can increase geological storage capacity, and wettability is the critical factor in the effectiveness of capillary trapping that limits CO2 escape from the pores of the target rock. ESal has state of the art knowledge in wettability and can help you increase the amount of CO2 stored underground, reduce the AOR to improve project economics, and significantly diminish the risk of CO2 escape.

ESal technology adds another seal to your carbon storage site
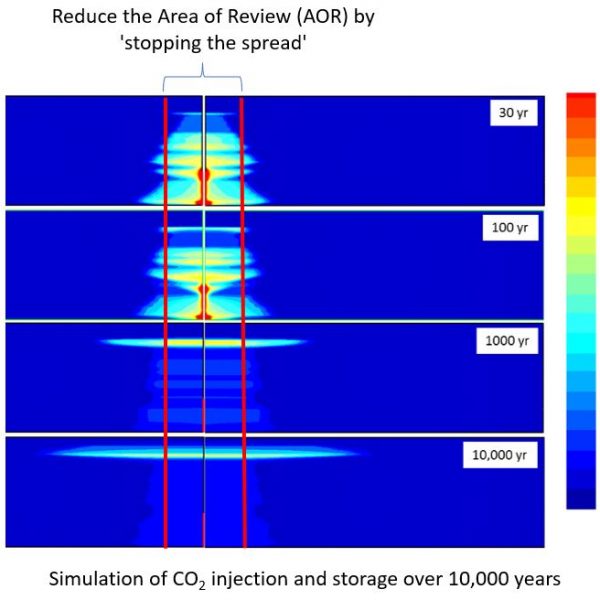
The challenge of CO2 storage is to contain as much CO2 in the smallest volume possible to reduce environmental risks. At ESal, we believe that carbon storage sites should not put future generations at risk. We are dedicated to leveraging our expertise and technology to improve the performance and safety of storage sites for the next 10,000 years.
Esal`s technology alters the wettability of storage sites to maximize CO2 retention while minimizing the potential for future leakage into our environment. When technology is implemented, we expect a smaller area of review, lower permitting costs, longer CO2 retention, add an extra layer of security, and minimizing risks for all stakeholders.
Increase capacity of reservoir pore space by altering wettability
Water chemistry changes rock wettability and is used to make storage sites safer by limiting CO2 mobility and trapping more CO2 in the same volume
Assess the current wettability of target storage formations and rank potential targets
Increase residual trapping by altering wettability, reducing the chance of CO2 escaping
Determine optimal technical and economic conditions for the project
Increased CO2 entrapment reduces the footprint during storage, significantly reducing associated permitting and maintenance costs
ESal screens storage sites, tests the target formations, and
designs the correct treatment to improve the storage sites’ capacity to safely store CO2
Customized Solutions
Frequently Asked Questions
CCS is the capture of CO2 that would otherwise be emitted to the atmosphere and the storage of that CO2 using a method that will store the capture CO2 for long periods of time to prevent the CO2 from returning to the atmosphere.
Carbon dioxide can be stored for significant periods of time in the deep ocean or underground (geological storage) as a supercritical liquid or by reacting the CO2 with other components to form a stable mineral.
There are four mechanisms that trap CO2 underground. They are 1) stratigraphic or structural traps, 2) solubility (dissolving CO2 in groundwater), 3) mineralization (formation of a stable mineral) and 4) capillary trapping which happens when the supercritical liquid is unable to move out of the pore spaces of the rock.
Supercritical CO2 is a liquid form of CO2 that is the natural state when there is sufficient pressure to change the gas into a liquid. Supercritical CO2 has a density about half that of water but has a low viscosity (very slippery). This is the standard form when large quantities of CO2 are transported by truck or pipeline.
The most important mechanism is capillary trapping.
Capillary trapping can store between 10% and 50% of the volume of the pore space and the amount is controlled by the wettability of the rock.
